计算溶液所需的质量、体积或浓度。
This is a demo store. No orders will be fulfilled.

| 货号 (SKU) | 包装规格 | 是否现货 | 价格 | 数量 |
|---|---|---|---|---|
| rp156087-10μg |
10μg |
现货  |
| |
| rp156087-100μg |
100μg |
现货  |
| |
| rp156087-500μg |
500μg |
期货  |
| |
| rp156087-1mg |
1mg |
期货  |
|
| 产品名称 | Recombinant Human IFN-alpha-2B Protein |
|---|---|
| 别名 | 重组人 IFN-alpha-2B 蛋白 |
| 英文别名 | IFN-alpha-2 | Interferon alpha-A | LeIF A |
| 规格或纯度 | 无动物源, Carrier Free, 生物活性, ActiBioPure™, ≥98%(SDS-PAGE&HPLC) |
| 产品介绍 |
干扰素(IFN)以其在宿主防御机制中的关键作用而闻名。Ⅰ型干扰素包括IFN-alpha 家族,IFN-beta, -omega,-kappa 和 Limitin/IFN-zeta,对调节病毒感染的免疫应答具有重要意义。Ⅱ型干扰素组仅包括干扰素γ。IFN-γ是一种多功能细胞因子,具有抗增殖、免疫调节和促炎症作用,对免疫应答的许多方面都很重要。Ⅲ型干扰素家族包括IL-29/IFN-lambda 1, IL-28A/IFN-lambda 2, IL-28B/IFN-lambda 3, and IFN-lambda 4。与Ⅰ型干扰素类似,Ⅲ型干扰素具有抗病毒、抗增殖和免疫调节作用。 IFN-αs are proteins secreted by leukocyte. They are mainly involved in innate immune response against viral infection. The IFN-α family has 13 subtypes and 23 different variants. The individual proteins have molecular masses between 19-26 kDa and consist of proteins with lengths of 156-166 and 172 amino acids. All IFN-α subtypes possess a common conserved sequence region between amino acid positions 115-151 while the amino-terminal ends are variable. Many IFN-alpha subtypes differ in their sequences at only one or two positions. Naturally occurring variants also include proteins truncated by 10 amino acids at the carboxy-terminal end. |
| 生物活性 | Fully biologically active when compared to standard. The specific activity determined by an anti-viral assay is no less than 1.6 × 10^8 IU/mg. |
| 内毒素水平 | <0.1 EU/μg |
| 表达系统 | 酵母 |
| 种属 | 人(Human) |
| 氨基酸 | 24-188 aa |
| 序列 | CDLPQTHSLG SRRTLMLLAQ MRRISLFSCL KDRHDFGFPQ EEFGNQFQKA ETIPVLHEMI QQIFNLFSTK DSSAAWDETL LDKFYTELYQ QLNDLEACVI QGVGVTETPL MKEDSILAVR KYFQRITLYL KEKKYSPCAW EVVRAEIMRS FSLSTNLQES LRSKE |
| 纯度 | >98% ( SDS-PAGE and HPLC) |
| 蛋白标签 | No tag |
| 蛋白长度 | Full length protein |
| 无载体 | Yes |
| 无动物源 | Yes |
| Accession # | P01563 |
| 来源 | 重组表达 |
| 预测分子量 | 19.3 kDa |
| 物理形态 | 冻干(Lyophilized) |
|---|---|
| 储存缓冲液 | Lyophilized from a 0.2 µm filtered solution in 1×PBS, pH 7.4, with 0.02 % Tween-20. |
| 复溶 | We recommend that this vial be briefly centrifuged prior to opening to bring the contents to the bottom. Reconstitute in sterile distilled water or aqueous buffer containing 0.1 % BSA to a concentration of 0.1-1.0 mg/mL. Stock solutions should be apportioned into working aliquots and stored at ≤ -20 °C. Further dilutions should be made in appropriate buffered solutions. |
| 储存温度 | -20°C储存,避免反复冻融 |
| 运输条件 | 超低温冰袋运输 |
| 稳定性与储存 | 12 months from date of receipt, -20 to -70 °C as supplied. 1 month, 2 to 8 °C under sterile conditions after reconstitution. 3 months, -20 to -70 °C under sterile conditions after reconstitution. Upon delivery aliquot. Avoid freeze/thaw cycle. |
| 分子类型 | 蛋白质 |

Recombinant Human IFN-alpha-2B Protein (rp156087)-Protein Bioactivity
Fully biologically active when compared to standard. The specific activity determined by an anti-viral assay is no less than 1.6×10⁸ IU/mg.
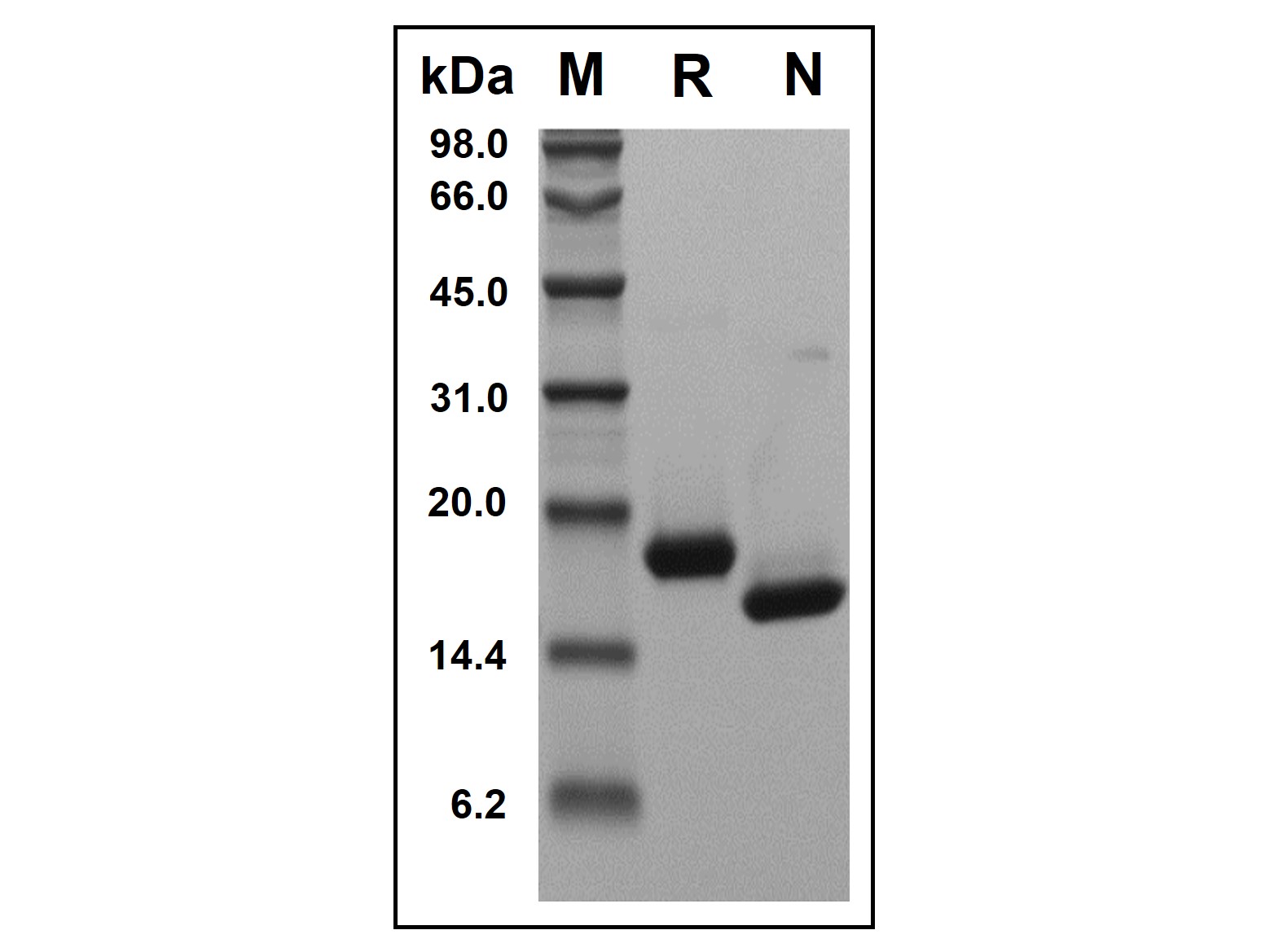
Recombinant Human IFN-alpha-2B Protein (rp156087)-SDS-PAGE
Recombinant Human IFN-alpha-2B Protein was resolved with SDS-PAGE under reducing (R) and non-reducing (N) conditions and visualized by Coomassie® Blue staining. Showing a single band at 19.3 kDa under reducing conditions and 16.2 kDa under non-reducing conditions.
通过匹配包装上的批号来查找并下载产品的 COA,每批产品都进行了严格的验证,您可放心使用!
| 批号(Lot Number) | 证书类型 | 货号 |
|---|---|---|
| 分析证书 | rp156087 | |
| 分析证书 | rp156087 | |
| 分析证书 | rp156087 | |
| 分析证书 | rp156087 |
| 1. Adolf, G R GR, Kalsner, I I, Ahorn, H H, Maurer-Fogy, I I and Cantell, K K.. (1991) Natural human interferon-alpha 2 is O-glycosylated.. The Biochemical journal, (1): [PMID:2049076] |
| 2. Oliver, G G, Balbás, P P, Valle, F F, Soberón, X X and Bolívar, F F.. [Cloning of human leukocyte interferon cDNA and a strategy for its production in E. coli].. Revista latinoamericana de microbiologia, [PMID:3906813] |
| 3. Streuli, M M, Nagata, S S and Weissmann, C C.. (1980) At least three human type alpha interferons: structure of alpha 2.. Science (New York, N.Y.), (19): [PMID:6158094] |
| 4. Allen, G G and Fantes, K H KH.. (1980) A family of structural genes for human lymphoblastoid (leukocyte-type) interferon.. Nature, (2): [PMID:6159537] |
| 5. Goeddel, D V DV and 17 more authors.. (1980) Human leukocyte interferon produced by E. coli is biologically active.. Nature, (2): [PMID:6159538] |
| 6. and Wetzel, R R.. (1981) Assignment of the disulphide bonds of leukocyte interferon.. Nature, (12): [PMID:6162107] |
| 7. Goeddel, D V DV and 9 more authors.. (1981) The structure of eight distinct cloned human leukocyte interferon cDNAs.. Nature, (5): [PMID:6163083] |
| 8. Lawn, R M RM and 5 more authors.. (1981) DNA sequence of a major human leukocyte interferon gene.. Proceedings of the National Academy of Sciences of the United States of America, [PMID:6170983] |
| 9. Weber, H H and Weissmann, C C.. (1983) Formation of genes coding for hybrid proteins by recombination between related, cloned genes in E. coli.. Nucleic acids research, (25): [PMID:6310510] |
| 10. Lee, N N and 7 more authors.. (1995) Interferon-alpha 2 variants in the human genome.. Journal of interferon & cytokine research : the official journal of the International Society for Interferon and Cytokine Research, [PMID:7627809] |
| 11. Murgolo, N J NJ and 9 more authors.. (1993) A homology model of human interferon alpha-2.. Proteins, [PMID:8234245] |
| 12. Radhakrishnan, R R and 6 more authors.. (1996) Zinc mediated dimer of human interferon-alpha 2b revealed by X-ray crystallography.. Structure (London, England : 1993), (15): [PMID:8994971] |
| 13. Klaus, W W, Gsell, B B, Labhardt, A M AM, Wipf, B B and Senn, H H.. (1997) The three-dimensional high resolution structure of human interferon alpha-2a determined by heteronuclear NMR spectroscopy in solution.. Journal of molecular biology, (12): [PMID:9417943] |
| 14. Nyman, T A TA, Tölö, H H, Parkkinen, J J and Kalkkinen, N N.. (1998) Identification of nine interferon-alpha subtypes produced by Sendai virus-induced human peripheral blood leucocytes.. The Biochemical journal, (15): [PMID:9425112] |
| 15. Austruy, E E and 7 more authors.. A defective retroviral vector encoding human interferon-alpha2 can transduce human leukemic cell lines.. Cancer gene therapy, [PMID:9694076] |
| 16. Humphray, S J SJ and 147 more authors.. (2004) DNA sequence and analysis of human chromosome 9.. Nature, (27): [PMID:15164053] |
| 17. Sjöblom, Tobias T and 28 more authors.. (2006) The consensus coding sequences of human breast and colorectal cancers.. Science (New York, N.Y.), (13): [PMID:16959974] |
| 18. Quadt-Akabayov, Sabine R SR, Chill, Jordan H JH, Levy, Rina R, Kessler, Naama N and Anglister, Jacob J.. (2006) Determination of the human type I interferon receptor binding site on human interferon-alpha2 by cross saturation and an NMR-based model of the complex.. Protein science : a publication of the Protein Society, [PMID:17001036] |
| 19. Gull, Iram I, Samra, Zahoor Qadir ZQ, Aslam, Muhammad Shahbaz MS and Athar, Muhammad Amin MA.. (2013) Heterologous expression, immunochemical and computational analysis of recombinant human interferon alpha 2b.. SpringerPlus, [PMID:23875128] |