This is a demo store. No orders will be fulfilled.
Protease Inhibitors
Proteases are widely present in cells and play a critical role in protein degradation and homeostasis regulation. However, during biochemical and molecular biology experiments, samples are susceptible to degradation by endogenous proteases during lysis or purification, which can lead to loss or functional impairment of target proteins. To prevent this, researchers typically add protease inhibitors to the lysis buffer to maintain sample integrity.
Ⅰ. Classification of Protease Inhibitors
Due to the diversity of proteases, there is no “universal” inhibitor. In practice, multiple inhibitors are often combined based on sample characteristics. Common inhibitors can be classified according to their targets:
1.Serine Protease Inhibitors
Targets: Trypsin, chymotrypsin, thrombin, and other serine proteases.
Representative Inhibitors:
- PMSF (Phenylmethylsulfonyl fluoride): A classic inhibitor with high efficiency, but extremely unstable in aqueous solution (half-life ~30 minutes), and should be added immediately before use.
- AEBSF (4-(2-Aminoethyl)benzenesulfonyl fluoride): Water-soluble with better stability and safety than PMSF.
- Aprotinin: A small protein that reversibly inhibits multiple serine proteases.
Name | ID | Grade & Purity |
PMSF Solution | 100mM in ethanol | |
Phenylmethanesulfonyl fluoride | Moligand™, ≥98%(GC) | |
PMSF | 10mM in DMSO | |
4-(2-Aminoethyl)benzenesulfonyl fluoride hydrochloride | 10mM in DMSO | |
AEBSF Solution | 50mg/ml in H2O | |
AEBSF Solution | 10mg/ml in H2O | |
4-(2-Aminoethyl)benzenesulfonyl fluoride hydrochloride | ≥98% | |
Recombinant Aprotinin | EnzymoPure™, 10mM in Water | |
Aprotinin bovine | Recombinant, ≥98%(SDS-PAGE), expressed in Nicotiana(tobacco),≥5 TIU/mg protein | |
Aprotinin from bovine lung | UltraBio™, ≥98%(SDS-PAGE), 3-8 TIU/mg solid | |
Aprotinin | EnzymoPure™, High-purity | |
Recombinant Aprotinin | EnzymoPure™, ≥3 EPU/mg | |
Aprotinin from bovine lung | EnzymoPure™, 3-8 TIU/mg |
2. Cysteine Protease Inhibitors
Targets: Cathepsins, such as Cathepsin B and L.
Representative Inhibitors:
- E64: Highly efficient, specific, irreversible, and stable.
- Leupeptin: A reversible inhibitor that also exhibits activity against some serine proteases.

Figure 1. Chemical structure of E-64
Name | ID | Grade & Purity |
E-64 | 10mM in DMSO | |
E-64 | Moligand™, ≥99%, protease inhibitor | |
Leupeptin | EnzymoPure™, 10mM in DMSO | |
Leupeptin | UltraBio™, ≥95%(HPLC), microbial | |
Leupeptin hemisulfate salt | Moligand™, ≥90%(HPLC) | |
Leupeptin | High-purity, EnzymoPure™ |
3. Aspartic Protease Inhibitors
Targets: Pepsin, Cathepsin D, renin.
Representative Inhibitor:
- Pepstatin A: Highly specific and effective at low concentrations, but poorly soluble; typically prepared in methanol or ethanol.
Name | ID | Grade & Purity |
Pepstatin | Moligand™, ≥75%(HPLC) | |
Pepstatin | 10mM in DMSO | |
Pepstatin A TFA | ≥97% |
4.Metalloprotease Inhibitors
Targets: Collagenases, thermolysin, and other Zn²⁺-dependent proteases.
Representative Inhibitors:
- EDTA / EGTA: Act by chelating metal ions; broad-spectrum and commonly used.
- 1,10-Phenanthroline: Also inhibits enzyme activity through metal ion chelation.
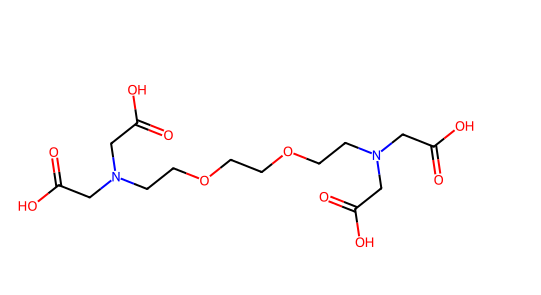
Figure 2. Chemical structure of EGTA
Name | ID | Grade & Purity |
EDTA disodium salt concentrate | Diluted to 1 liters, the diluted concentration was 0.1M | |
EDTA disodium salt concentrate | Diluted to 1 liters, the diluted concentration was 0.01M | |
Ethylenediaminetetraacetic acid disodium salt dihydrate | UltraBio™, Ultra pure, ≥98.5% | |
Ethylene glycol-bis(2-aminoethylether)-N,N,N′,N′-tetraacetic acid (EGTA) | AR, ≥99% | |
Ethylene glycol-bis(2-aminoethylether)-N,N,N′,N′-tetraacetic acid | Suitable for molecular biology, ≥99% | |
1,10-Phenanthroline Monohydrate | 10mM in DMSO | |
1,10-Phenanthroline Monohydrate | ACS, ≥99% | |
1,10-Phenanthroline Monohydrate | AR, ≥98% |
5.Aminopeptidase Inhibitors
Targets: Proteases that sequentially cleave amino acids from the N-terminus of peptide chains.
Representative Inhibitors:
- Bestatin: Effective against aminopeptidase B and leucine aminopeptidase.
- EDTA: Also inhibits metal-dependent aminopeptidases.
Name | ID | Grade & Purity |
Bestatin hydrochloride | ≥98%(HPLC) |
6.Proteasome Inhibitors
Targets: The 26S proteasome complex.
Representative Inhibitors:
- MG132: A reversible inhibitor commonly used to study the ubiquitin–proteasome pathway.
- Lactacystin: Irreversible and highly specific.

Figure3. Chemical structure of MG-132
Name | ID | Grade & Purity |
MG-132 | Moligand™, ≥98% | |
Lactacystin | BioReagent, ≥95% |
II. Usage Strategies
1. Principle of Combined Use
Tissue and cell samples often contain multiple types of proteases. Therefore, it is recommended to use a mixture of inhibitors rather than a single inhibitor to achieve broad-spectrum protection.
2. Sources and Preparation
- Commercially available inhibitor cocktails: Typically provided as tablets or ready-to-use solutions, with optimized ratios. Convenient for routine experiments.
- Custom preparation: Inhibitors can be combined according to experimental needs. Below is a basic formulation for protein extraction from mammalian cells or tissues (working concentrations for reference only).
Inhibitor | Working Concentration | Target Protease Type | Stock Solution (Solvent) | Storage Condition | Catalog Number |
PMSF | 1 mM | Serine proteases | 100 mM (ethanol / isopropanol) | –20 °C, protected from light | |
Aprotinin | 0.31 μM | Serine proteases | 1 mg/mL (deionized water) | –20 °C, aliquoted | |
Leupeptin | 0.5 μg/mL | Cysteine + Serine proteases | 1 mg/mL (deionized water) | –20 °C, aliquoted | |
Pepstatin A | 1 μM | Aspartic proteases | 1 mg/mL (methanol / ethanol) | –20 °C, protected from light | |
EDTA | 0.5 mM | Metalloproteases | 0.5 M (deionized water, pH adjusted to 8.0) | Room temperature |
Note:PMSF must be freshly added before use. Stock solutions should be aliquoted and stored at –20 °C.
III. Experimental Considerations
1.Low-temperature handling: Perform all steps on ice to reduce protease activity.
2.Pre-addition of inhibitors: Add inhibitors to the lysis buffer before use to ensure immediate protection of the sample.
3.Concentration control: Avoid excessive use, as this may interfere with downstream experiments (e.g., immunoprecipitation, functional assays).
4.Stability differences: Some inhibitors (e.g., PMSF) must be used quickly to prevent inactivation.
5.Special requirements: For studies involving phosphorylated proteins, additional phosphatase inhibitors (e.g., NaF, β-glycerophosphate, Na₃VO₄) should be included.
6.Safety precautions: Many inhibitors are toxic; handle them in a fume hood and wear appropriate protective equipment.
Aladdin: https://www.aladdinsci.com/
 首页
首页 400-620-6333
400-620-6333
