This is a demo store. No orders will be fulfilled.
PMHS, Polymethylhydrosiloxane
Product Manager
Sandra Forbes

Polymethylhydrosiloxane (PMHS), a byproduct generated within the silicone manufacturing sector, serves as an economical, user-friendly, and eco-conscious reducing agent. Notably, PMHS exhibits superior stability in the presence of air and moisture compared to alternative silanes, enabling prolonged storage without any compromise in its activity.

![]()
Recent Literature
![]()

Stannous chloride facilitates a chemoselective reductive amination process, enabling the reaction of diverse carbonyl compounds with aromatic amines. This method employs an inexpensive and environmentally benign reducing agent, polymethylhydrosiloxane (PMHS), in methanol as the solvent. The approach is versatile, suitable for the synthesis of both tertiary and secondary amines.
O. S. Nayal, V. Bhatt, S. Sharma, N. Kumar, J. Org. Chem., 2015, 80, 5912-5918.
![]()

An efficient catalytic system has been established for the reductive alkylation of secondary amines with aldehydes, utilizing an iridium complex as the catalyst and Et₃SiH as the reducing agent. Additionally, a more economical, user-friendly, and eco-conscious alternative, polymethylhydrosiloxane (PMHS), was demonstrated to be equally effective in place of Et₃SiH.
T. Mizuta, S. Sakaguchi, Y. Ishii, J. Org. Chem., 2005, 70, 2195-2199.
DOI: 10.1021/jo0481708
![]()

Transition-metal-free catalytic strategies have been developed for the controlled reduction of amide functionalities. These methods utilize affordable and bench-stable hydrosilanes as reducing agents, enabling the selective reduction of unactivated C–O bonds in amides. By adjusting the hydrosilane and solvent, the C–N bond can be selectively cleaved through a deacylative process.
W. Yao, L. He, D. Han, A. Zhong, J. Org. Chem., 2019, 84, 14627-14635.
![]()

Copper N-heterocyclic carbene complexes act as dual catalysts, promoting both the aerobic oxidation of alcohols to aldehydes and the reduction of imines to amines. A one-pot tandem synthetic approach enables the conversion of benzylic alcohols and anilines into valuable secondary amines through an oxidation-reduction sequence.
L.-W. Zhan, L. Han, P. Xing, B. Jiang, Org. Lett., 2015, 17, 5990-5993.
DOI: 10.1021/acs.orglett.5b02756
![]()

The activation of PMHS by KF allows for the efficient recycling of Me₃SnH during a Pd(0)-catalyzed hydrostannation/Stille cascade reaction. This protocol is characterized by short reaction times and proceeds via a Me₃SnF intermediate.
R. E. Maleczka, W. P. Gallagher, Org. Lett., 2001, 3, 4173-4176.
DOI: 10.1021/ol016792z
![]()

The copper-catalyzed ring-opening hydroamination of methylenecyclopropanes with polymethylhydrosiloxane and O-benzoylhydroxylamines yields homoallylamines in excellent yields. This reaction proceeds with selective cleavage of the cyclopropane C–C bond at the more sterically hindered, proximal position.
D. Nishikawa, R. Sakae, Y. Miki, K. Hirano, M. Miura, J. Org. Chem., 2016, 81, 12128-12134.
![]()

An efficient, directed reductive amination protocol for β-hydroxy-ketones enables the stereoselective synthesis of 1,3-syn-amino alcohols. This method employs Ti(iOPr)₄ for the coordination of the intermediate imino alcohol and polymethylhydrosiloxane (PMHS) as the reducing agent.
D. Menche, F. Arikan, J. Li, S. Rudolph, Org. Lett., 2007, 9, 267-270.
DOI: 10.1021/ol062715y
![]()

A copper-catalyzed hydrocarbonylative C–C coupling reaction between unactivated alkyl iodides and terminal alkynes facilitates the highly chemo- and regioselective synthesis of unsymmetrical dialkyl ketones. This method exhibits broad functional group tolerance and is effective with both primary and secondary alkyl iodides.
L.-J. Cheng, N. P. Mankad, J. Am. Chem. Soc., 2017, 139, 10200-10203.
DOI: 10.1021/jacs.7b05205
![]()

The palladium-catalyzed reduction of aromatic nitro groups to amines can be achieved in high yields, with broad functional group compatibility and short reaction times at room temperature, using an aqueous potassium fluoride and polymethylhydrosiloxane (PMHS) system. In contrast, aliphatic nitro compounds are reduced to the corresponding hydroxylamines using triethylsilane instead of the PMHS/KF combination.
R. J. Rahaim, R. E. Maleczka, Jr., Org. Lett., 2005, 7, 5087-5090.
DOI: 10.1021/ol052120n
![]()

Ketones can be efficiently reduced to the corresponding methylene compounds using a convenient and cost-effective combination of polymethylhydrosiloxane (PMHS) and FeCl₃.
C. Dal Zotto, D. Virieux, J.-M. Campagne, Synlett, 2009, 276-278.
![]()

A catalytic system comprising Pd(OAc)₂ and polymethylhydrosiloxane (PMHS) facilitates the chemo-, regio-, and stereoselective deoxygenation of benzylic oxygenated substrates in the presence of aqueous KF and a catalytic amount of an aromatic chloride. This process involves palladium-nanoparticle-catalyzed hydrosilylation followed by C–O bond reduction. The chloroarene promotes hydrogenolysis through the slow, controlled release of HCl.
R. J. Rahaim, Jr., R. E. Maleczka, Jr., Org. Lett., 2011, 13, 584-587.
DOI: 10.1021/ol102757v
![]()

A tandem catalytic system combining heterogeneous Pd/TiO₂ and homogeneous FeCl₃ enables a rapid and practical protocol for the chemoselective deoxygenation of various aromatic ketones and aldehydes. This method employs polymethylhydrosiloxane (PMHS) as a green hydrogen source.
Z. Dong, J. Yuan, Y. Xiao, P. Mao, W. Wang, J. Org. Chem., 2018, 83, 11067-11073.
![]()

Diethylzinc (Et₂Zn) serves as an efficient and chemoselective catalyst for the reduction of tertiary amides under mild reaction conditions, utilizing polymeric silane (PMHS) as a cost-effective hydride source. The addition of a substoichiometric amount of lithium chloride is crucial for catalytic activity. A range of amides, including those with additional functional groups, were reduced to their corresponding amines in excellent yields.
O. O. Kovalenko, A. Volkov, H. Adolfsson, Org. Lett., 2015, 17, 446-449.
DOI: 10.1021/ol503430t
![]()

An economical, ligand-modified version of Stryker's reagent, based on a bidentate, achiral bis-phosphine, is generated in situ. This " (BDP)CuH " complex effectively catalyzes the conjugate reduction of various unsaturated substrates, including those that are typically unreactive toward Stryker's reagent.
B. A. Baker, Ž. V. Bošković, B. H. Lipshutz, Org. Lett., 2008, 10, 289-292.
DOI: 10.1021/ol702689v
![]()

A highly chemoselective conjugate reduction of electron-deficient Michael acceptors, including α,β-unsaturated ketones, carboxylic esters, nitriles, and nitro compounds, is achieved using PMHS in the presence of a catalytic amount of B(C₆F₅)₃.
S. Chandrasekhar, G. Chandrasekhar, M. S. Reddy, P. Srihari, Org. Biomol. Chem., 2006, 4, 1650-1652.
DOI: 10.1039/b603610b
![]()

Leveraging micellar catalysis in water, asymmetric hydrosilylation reactions can be conducted at ambient temperatures using water as the global reaction medium.
S. Huang, K. R. Voigtritter, H. B. Unger, B. H. Lipshutz, Synlett, 2010, 2041-2044.
![]()

A catalytic complex of CuH with a nonracemic JOSIPHOS or SEGPHOS ligand enables highly efficient and enantioselective 1,4-reductions of α,β-disubstituted enoates and lactones, using PMHS as the stoichiometric reducing agent.
B. H. Lipshutz, J. M. Servesko, B. R. Taft, J. Am. Chem. Soc., 2004, 126, 8352-8353.
DOI: 10.1021/ja049135l
![]()

A copper-catalyzed reduction of aryl/heteroaryl ketones, employing Cu(OAc)₂·H₂O and (R)-3,4,5-MeO-MeO-BIPHEP as the ligand, provides nonracemic secondary alcohols in excellent yields and with high enantiomeric excess (ee) values. This reaction is conducted in an aqueous micellar medium using PMHS as an inexpensive, benign, and convenient stoichiometric hydride source.
D. M. Fialho, E. Etemadi-Davan, O. C. Langner, B. S. Takale, A. Gadakh, G. Sambasivam, B. H. Lipshutz, Org. Lett., 2021, 23, 3283-3286.
DOI: 10.1021/acs.orglett.1c00746
![]()

A complex of CuH with Takasago's nonracemic ligand, DTBM-SEGPHOS, is a highly reactive reagent for the asymmetric hydrosilylation of heteroaromatic ketones under very mild conditions. PMHS serves as an inexpensive source of hydride for the in situ generation of CuH.
B. H. Lipshutz, A. Lower, K. Noson, Org. Lett., 2002, 4, 4045-4048.
DOI: 10.1021/ol026755n
![]()

The use of (R)-(−)-(DTBM-SEGPHOS)CuH facilitates a highly enantioselective 1,2-hydrosilylation of prochiral diaryl ketones, yielding nonracemic diarylmethanols in excellent yields.
C.-T. Lee, B. H. Lipshutz, Org. Lett., 2008, 10, 4187-4190.
DOI: 10.1021/ol801590j
![]()

In situ generation of an extremely reactive copper hydride species, ligated by a nonracemic SEGPHOS ligand, enables highly enantioselective asymmetric hydrosilylations of conjugated cyclic enones.
B. H. Lipshutz, J. M. Servesko, T. B. Petersen, P. P. Papa, A. A. Lover, Org. Lett., 2004, 6, 1273-1275.
DOI: 10.1021/ol0400185
![]()

An efficient semireduction of alkynes, including both internal and terminal alkynes, can be achieved using small amounts of a copper catalyst without over-reduction. This new method exhibits excellent chemoselectivity and tolerates nitro and aryl iodo groups. The commercial availability of the catalyst precursor further enhances the appeal of this catalytic system.
A. M Whittaker, G. Lalic, Org. Lett., 2013, 15, 1112-1115.
DOI: 10.1021/ol4001679

![]()
A. M Whittaker, G. Lalic, Org. Lett., 2013, 15, 1112-1115.
DOI: 10.1021/ol4001679
![]()

The hydroallylation of terminal alkyl and aryl alkynes with simple allyl phosphates and 2-substituted allyl phosphates yields skipped dienes. Additionally, the hydroallylation of functionalized internal alkynes facilitates the synthesis of complex trisubstituted alkenes.
M. Mailig, A. Hazra, M. K. Armstrong, G. Lalic, J. Am. Chem. Soc., 2017, 139, 6969-6977.
DOI: 10.1021/jacs.7b02104
![]()

A copper-catalyzed regio- and enantioselective hydroallylation of alkenyl boronates and boramides with allylic phosphates, in the presence of hydrosilanes, enables the efficient synthesis of a broad range of homoallylic alkylboron compounds with good yields and high enantioselectivities.
J. T. Han, W. J. Jang, N. Kim, J. Yun, J. Am. Chem. Soc., 2016, 138, 15146-15149.
DOI: 10.1021/jacs.6b11229
![]()

In the presence of an IMes-Cu catalyst, a tandem hydrocupration/allylation of alkenyl boronates with allyl phosphate proceeds efficiently in the presence of a hydrosilane via in situ generated β-α-copper intermediates. Mono- and disubstituted alkenyl boronates react effectively with terminal allyl phosphates, while trisubstituted substrates exhibit limited reactivity.
W. J. Jang, J. T. Han, J. Yun, Synthesis, 2017, 49, 4753-4758.
![]()

A copper hydride-catalyzed Sₙ²'-reduction of propargylic carbonates yields functionalized allenes in good yields. This method benefits from the stabilizing effect of NHC ligands on CuH, offering high reactivity, stereoselectivity, and functional group tolerance.
C. Deutsch, B. H. Lipshutz, N. Krause, Org. Lett., 2009, 11, 5010-5012.
DOI: 10.1021/ol901868m
![]()

A highly enantioselective reduction of α,β-unsaturated nitriles can be achieved using a Cu(OAc)₂/josiphos complex as the catalyst under hydrosilylation conditions. This reaction provides access to valuable β-aryl-substituted chiral nitriles with good yields and excellent enantioselectivities.
D. Lee, D. Kim, S. Yun, Angew. Chem. Int. Ed., 2006, 45, 2785-2787.
![]()

A range of 3-aryl-3-pyridylacrylonitriles can be reduced with high enantioselectivity under optimal conditions using a copper/Josiphos complex in the presence of polymethylhydrosiloxane (PMHS).
D. Lee, Y. Yang, J. Yun, Org. Lett., 2007, 9, 2749-2751.
DOI: 10.1021/ol0712071
![]()

Deprotection of allyl ethers, amines, and esters to release hydroxyl, amino, and acid groups can be achieved under mild conditions using a reagent combination of polymethylhydrosiloxane (PMHS), ZnCl₂, and Pd(PPh₃)₄.
S. Chandrasekhar, R. Reddy, R. J. Rao, Tetrahedron, 2001, 57, 3435-3438.
DOI: 10.1016/S0040-4020(01)00187-9
![]()
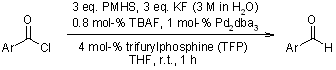
Under Pd(0) catalysis, polymethylhydrosiloxane (PMHS) can efficiently reduce aryl acid chlorides to their corresponding aldehydes in the presence of fluoride, without requiring an additional reductant.
K. Lee, R. E. Maleczka, Jr., Org. Lett., 2006, 8, 1887-1888.
DOI: 10.1021/ol060463v
![]()

An efficient palladium-catalyzed reduction of N-(tert-butoxycarbonyl)indoles yields N-(tert-butoxycarbonyl)indolines in good yields at room temperature, using polymethylhydrosiloxane (PMHS) as the reducing agent.
S. Chandrasekhar, D. Basu, C. R. Reddy, Synthesis, 2007, 1509-1512.
![]()
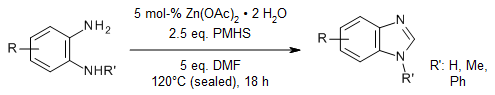
The use of various o-phenylenediamines and N-substituted formamides as C₁ sources in a zinc-catalyzed cyclization, in the presence of poly(methylhydrosiloxane), provides benzimidazoles in good yields. Benzoxazole and benzothiazole derivatives can also be synthesized using this method.
D. B. Nale, B. M. Bhanage, Synlett, 2015, 26, 2831-2834.
![]()

A silane-promoted nickel-catalyzed amination of aryl chlorides, using a catalytic amount of Ni(acac)₂ and 3,5,6,8-tetrabromo-1,10-phenanthroline as the ligand in the presence of polymethylhydrosiloxane, yields the desired (het)arylamines in good yields. The reaction is sensitive to the nature and amount of the silane promoter.
G. Manolikakes, A. Gavryushin, P. Knochel, J. Org. Chem., 2008, 73, 1429-1434.
DOI: 10.1021/jo702219f
![]()

A nickel-catalyzed enantioselective hydrofluoromethylation of enamides and enol esters, using ICH₂F as the fluoromethyl source, provides chiral α-fluoromethylated amides and esters with wide functional group compatibility and excellent enantioselectivity.
Y. Du, S. Chen, H. Cao, Y. Zhang, H. Lei, G. Xia, H. Huang, Z. Li, Org. Lett., 2023, 25, 2218-2222.
DOI: 10.1021/acs.orglett.3c00357
Quoted from: https://www.organic-chemistry.org/chemicals/reductions/polymethylhydrosiloxane-pmhs.shtm
Aladdinsci: https://www.aladdinsci.com
 首页
首页 400-620-6333
400-620-6333
